Fluorescence vs Phosphorescence – a Photographic Investigation
Fluorescence and phosphorescence are forms of luminescence, the emission of light without heat. The source of the energy for the emitted light is electromagnetic radiation (for our purposes, light) that shines on the material. Both processes start with absorption of an incoming photon, moving an electron to a higher orbital, an ‘excited state’. They end with that electron returning to the ground state, emitting a new photon of light in the process. At the physics (quantum mechanics) level the two phenomena differ in how that excited-state electron transitions back to the ground state. In fluorescence the transition is from the first excited state to the ground state and it happens fast – on a time scale on the order of 10 nanoseconds (10 billionths of a second) after absorption of the incoming photon. In phosphorescence the transition is from a so-called ‘forbidden state’ and the time scale can range from 100s of nanoseconds, to seconds, minutes, or even hours.
At a practical level, phosphorescence is behind things we call ‘glow-in-the-dark’ that are charged up by light and then continue to glow after the light source is removed. With fluorescence the emission stops essentially instantaneously as soon as the light source is removed.
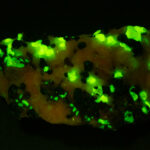
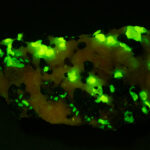
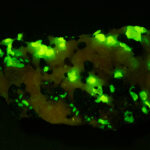
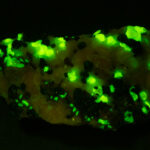
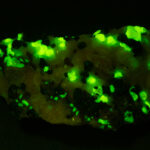
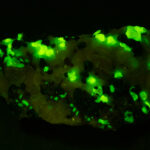
Given that background, I became intrigued by a seemingly odd photographic observation of a mineral specimen that appeared to be fluorescent – there was no glow-in-the-dark effect that I could see. This was a specimen containing calcite and willemite that came from the Sterling Hill Mine, in an area of New Jersey that is especially rich in fluorescent minerals.
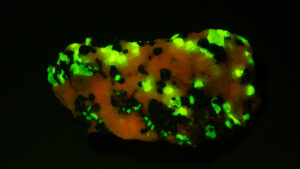
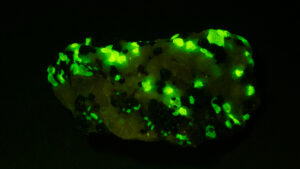
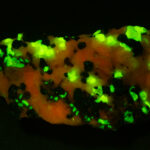
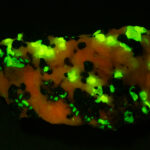
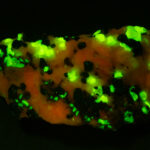
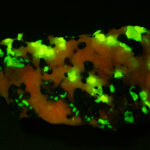
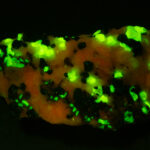
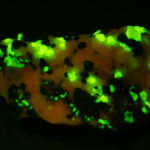
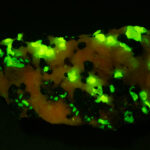
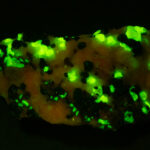
I first looked at the specimen with blue light excitation (not UV!) (see links at end of article) and could see characteristic green fluorescence from the willemite and red fluorescence (or so I thought) from the calcite, as in the center picture below. I then tried to photograph it using a portable camera setup with an electronic flash fitted with a blue-light excitation filter. The image I got was the one below right – clear willemite, no calcite. I was a bit perplexed since the camera setup always did a good job of capturing the fluorescence I could see.
(Click any image for larger view)
- White light
- Steady
- Flash
Back in the lab I set up in a dark room and took a picture of the specimen using a blue flashlight instead of the electronic flash. With this steady light source the exposure time was about ½ second and the image came out just the same as my eye saw it with nice green and red. I switched back to the flash and again, no red. But I noticed that when the flash fired I could see both green and red by eye.
When shooting with the flash I typically have the digital camera set on a very short exposure time, typically 1/200 of a second (5 milliseconds (thousandths of a second)). The duration of a flash is only about 2 milliseconds, so the 5 milliseconds should have been plenty of time to capture fluorescence excited by the flash. The short exposure time helps make a crisp image even with the camera hand-held rather than on a tripod.
I wondered if the time might be the factor so I took the flash photo again with a 1/2 second exposure and the camera mounted on a tripod in a dark room, the same as for the steady light image. This time the red showed up in the image and looked exactly like the photo taken with the flashlight. I then started to systematically reduce the exposure time bit by bit, as you can see in the photo sequence below. (You can enlarge the images by clicking on one and then step through with the left-right arrow buttons.)
- 1/2 second
- 1/4 second
- 1/5 second
- 1/8 second
- 1/10 second
- 1/15 second
- 1/20 second
- 1/30 second
- 1/40 second
- 1/50 second
- 1/60 second
- 1/80 second
- 1/100 second
- 1/125 second
- 1/160 second
- 1/200 second
As you sequence through the images in order of decreasing exposure time (from top left to bottom right) you can see the change in intensity of the red light. The green, a true fluorescence, stays constant. There is a slight diminution of the red intensity when you go from 1/10 to 1/15 second exposure and from then on you can see the intensity dropping steadily until it is effectively gone at 1/160 of a second. From this we can see that the red light is only starting to be emitted about 6 thousandths of a second after the flash fires, and it is on the order of 1/15 – 1/10 of a second (65 – 100 milliseconds) before all the light that is going to come out has been emitted.
The conclusion is that the red light emission from this calcite specimen is a true phosporescence, not a fluorescence, with an emission that does not end until about a tenth of a second after the excitation light has been removed. This is on the order of a million times slower than a true fluorescence!
Links