Fluorescence Excitation and Emission – Maxima vs Spectra
The purpose of this article is to foster understanding of the difference between excitation and emission maxima and excitation and emission spectra. One must be familiar with the nature of fluorescence to know why this distinction is important. It is not uncommon that we receive questions along the lines of ‘Do you have a light source at 488nm?’
The question itself tells us several things about the person who has inquired:
- They are working with GFP (green-fluorescent protein);
- They have consulted a colleague or a printed table that tells them that the excitation/emission values for GFP are 488/508 nm;
- They know that they need to use fluorescence, but they don’t really know the details of how it works;
- They have learned that NIGHTSEA provides fluorescence solutions, and they want to know if we have a solution for their particular application.
There is nothing wrong with the question. You can use fluorescence without understanding it. If you need to select equipment to work with fluorescence it is perfectly logical to consult references for guidance and then ask for what those references tell you. The 488/508nm that you will find for GFP is correct – those are the ‘best’ wavelengths at which to excite the fluorescence and to view the emitted light. What the references rarely tall you is that those are not the ONLY wavelengths for either excitation or emission.
We’re not going to go into the physics of fluorescence here. We do that in another article on this web site. The most relevant point in regard to how fluorescence works is that there is no single ‘magic’ wavelength for either fluorescence excitation or emission. A fluorescent subject can be excited at a range (or even more than one range!) of wavelengths and the resultant emission extends over a range of wavelengths. In each case the range is called a spectrum – the excitation spectrum and the emission spectrum.
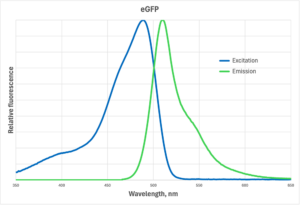
As an example, the curves in the graph below show the excitation and emission spectra for eGFP (enhanced GFP), the most commonly used fluorescent protein. The blue curve is the excitation spectrum and the green curve is the emission spectrum. The peaks are at 488nm and 508nm, exactly where the tables tell you they should be. 488nm is the most efficient wavelength on a watt for watt basis, but it is certainly not the only wavelength that can excite the fluorescence.
(Click image for larger view)
You can think of the excitation spectrum as a graph of the relative efficiency of various wavelengths of light for exciting fluorescence of the subject. If 100 units of light at 488nm produce x intensity of fluorescence, it would take about 200 units of light at 450nm to produce the same intensity, because 450nm light is about half as likely as 488nm light to be absorbed and result in fluorescence. But that doesn’t mean that you can’t use 450nm light!
The situation with the emission spectra is a bit different, in that you don’t get to choose what wavelengths come out. The nature of fluorescence from a particular fluorophore is that the emission spectrum is independent of the excitation wavelength. That is, the emitted light will be distributed over the same emission spectrum whether you excite at 440, 450, 460nm, etc.
The excitation spectrum comes into play in selecting the light source you choose to excite the fluorescence. The emission spectrum is useful in understanding the role of the barrier filter.
In an ideal system:
- The excitation light source would do the best possible job of exciting the fluorescence
- The emission barrier filter would do the best possible job of transmitting the emitted light
- The light source and barrier filter work together to eliminate any ‘crosstalk’ – excitation light passing through the barrier filter.
In real-world systems there generally need to be some trade-offs related to the spectral characteristics – the excitation and emission spectra – and the properties of light sources and filters. The last bullet point above is critical – if too much (or any!) excitation light gets through the barrier filter it can seriously degrade your ability to see the desired fluorescence, especially if that fluorescence is not very strong.
A good part of the challenge is due to the fact that the excitation and emission maxima for the fluorophores used in many applications, such as GFP, are very close together. This makes it challenging to pair light sources and filters that serve all three of the bullet points simultaneously, especially if you are trying to put together a robust system at a reasonable price point.
In order to meet all of these requirements the NIGHTSEA light sources are often exciting on the ‘shoulder’ of the excitation spectrum, rather than at the peak. Moving away from the peak provides more ‘room’ to select a barrier filter that (a) transmits the fluorescence emission effectively and (b) blocks any reflected excitation light. In the case of GFP, the NIGHTSEA Royal Blue (RB) source emits primarily in the 440-460nm range, a range that the graph shows is capable of exciting GFP fluorescence, while the paired excitation filters (either a longpass with cutoff at about 500nm or the bandpass (500 – 560nm)) transmit the emission well while doing a great job of blocking reflected light, achieving good viewing contrast.
The bottom line for any fluorescence application is – ‘can you see what you want to see?’ Our recommendations for NIGHTSEA wavelength set selection come either from direct customer experience with the listed fluorophores or from examination of full excitation and emission spectra. There are several ways that you can find spectra on your own:
- Spectra viewers
- Data sheets associated with products
- Do a web search for ‘xxx fluorescence excitation and emission’, where ‘xxx’ is the name of your fluorophore.
That’s what we do at NIGHTSEA to provide advice whenever we are asked about a new fluorophore.
We hope this article helps, and of course contact us if you have any questions.