Why NIGHTSEA Offers Different Excitation Wavelengths
We are sometimes asked why there are different excitation wavelengths, or questions along the lines of ‘What are the different excitation wavelengths for?’. The simple answer is:
You need multiple options for excitation wavelengths because different materials may need different wavelengths to cause them to fluoresce.
If that last sentence tells you what you need to know, you can stop there. For more explanation read on.
This results from the physics of fluorescence. For those who want to go into more depth on this you can look at our article ‘Physics of Fluorescence – the Jablonski Diagram‘, but what it really comes down to is that a fluorescing material can be characterized by excitation and emission spectra. The excitation spectrum is a plot of the relative efficiency of different wavelengths of light to excite fluorescence in the subject, while the emission spectrum is a plot of the relative distribution of energy released in the form of fluorescence. That is, fluorescing materials absorb light most effectively in some range of wavelengths and emit light in some other range of wavelengths.
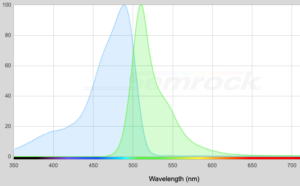
The goal in choosing a light source for an application is to select an option that will produce strong fluorescence in the sample being observed. Let’s look at a common example in the life sciences, Enhanced Green Fluorescent Protein (eGFP) as an example. In the graph below the curve shaded in blue is the excitation spectrum and the green-shaded curved is the emission spectrum. The blue curve tells you that wavelengths in the 450 – 500nm range should be good at producing fluorescence in the eGFP.
(Click image for larger view)
Our recommendation for observing eGFP is our Royal Blue (RB) light source, which emits in the 440 – 460nm range. You might think that the Cyan (CY) (490 – 515nm) would be better because it hits closer to the peak, but there is more to it than this. We address that in this article. The point is that not just any wavelength will be good for exciting eGFP. An Ultraviolet (UV) light, which emits at wavelengths shorter than 400nm, would not be effective. Similarly, the Violet (VI) light source, which emits in the 400 – 415nm range, would be a poor choice.
How do you know what wavelength range will make something fluoresce? Some options:
- For materials used in the life sciences, there are a number of online spectra viewers available.
- Article on wavelength choice on this web site
- Data sheets associated with products
- Manufacturer recommendations
Be careful of tables that just list excitation and emission wavelength maxima, usually in the format 488/508nm (the peaks for eGFP). They are correct about the peak, but they don’t tell you anything about the range of wavelengths that can work well. You do not have to be right on the peak to get good fluorescence! Sometimes the best approach is to try several options to see what works best. If your work involves multiple different fluorescing materials it can be helpful to have several different wavelength sets on hand for that.